On June 27, 2022, the journal Nature Methods published online the results of the latest research work of Hao Haiping/Ye Hui team (Cyclic immonium ion of lactyllysine reveals widespread lactylation in the human proteome). Wan Ning, a 2019 PhD student, and Wang Nian, a 2018 MS student, are the co-first authors of this paper, and our Distinguished Research Fellow Hui Ye, Professor Haiping Hao, and Professor Nanxi Wang of Nanjing University of Traditional Chinese Medicine are the co-corresponding authors of this paper, and China Pharmaceutical University is the first correspondent of this paper. The main collaborators of this work include academician Guangji Wang, Dr. Chang Shao and Siqin Yu, Hanqing Zhang, Dexiang Wang, Wenjie Lu, Ying Kong, Xinmiao Wang and Longlang Lu.
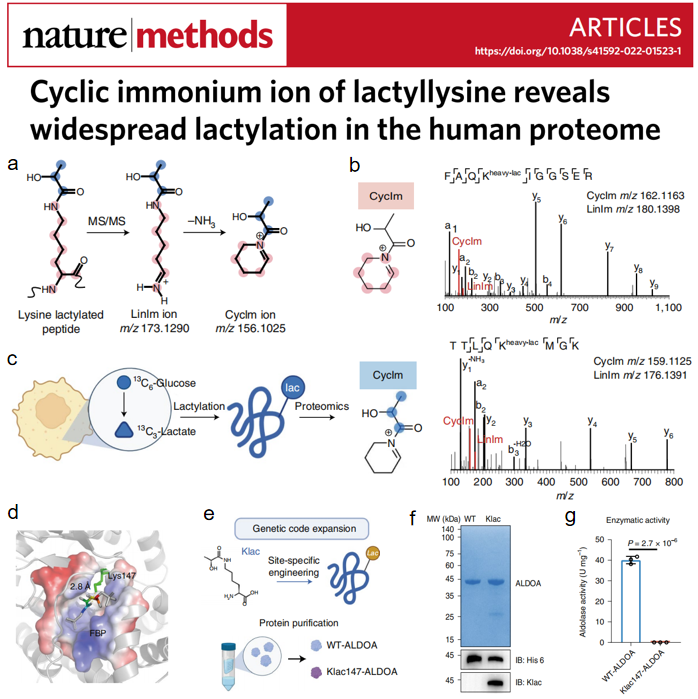
This work addresses the scientific question of whether lactate can directly covalently modify non-histone proteins and thus exert biological effects, and proposes a strategy to search for new substrate proteins modified by lactylation in public human proteome deep sequencing data. However, since the false-positive rate of retrieving modifications from non-enriched proteomic data is extremely high, the discovery of modification-specific signature ions can significantly reduce the false-positive rate of identification through spectroscopic screening, reveal the true modification sites and target proteins, and guide the subsequent biological validation. Based on this need, the team synthesized and studied the spectra of model lactylated peptides, and discovered for the first time that peptides carrying lactylated modified lysines form chain imide ions after secondary breakage in collision chambers, and the ions undergo deamination to form secondary fragments, cyclic imide ions. Further, by analyzing a large number of lactylation-positive peptides enriched in biological samples, and then using nearly 100,000 non-modified peptide spectra from the human proteome as a negative control, the sensitivity and specificity of the cyclic imide ion to indicate lactylation modification was confirmed, and the ion was proposed as the gold standard for determining the authenticity of lactylation retrieved from the database.
Based on this diagnostic ion strategy, the investigators mined the existing non-enriched, large-scale human proteomic data resources for novel, rich information on lactylation modification substrate proteins and loci, especially from the human cellular proteomic thermal stability data of the Meltome Atlas library published in Nature Methods 2020, and found that lactylation modifications are highly enriched in glycolytic pathway. Among them, the lactylation modification of metabolic enzyme ALDOA was present in a variety of human tumor cell lines with high occupancy ratio of the modification, which triggered the speculation that lactylation modification regulates metabolic enzyme activity and other functions, and thus regulates glycolytic pathway.
Hao Haiping and Ye Hui's group further collaborated with Wang Nanxi's group to achieve the first lactylation modification to the target protein ALDOA by using advanced chemical biology technology - gene codon expansion technology, and found that the enzyme activity was significantly reduced after the modification, revealing that lactate accumulation can covalently modify the upstream of glycolytic pathway and inhibit glycolysis by This study reveals that lactate accumulation can covalently modify the upstream metabolic enzymes of the glycolytic pathway and inhibit the activity of glycolysis, which complements the existing end-product inhibition regulatory model in biochemistry.
In summary, this study demonstrates that lactylation is a non-histone specific, biologically functional post-translational modification that is widely present in human tissues and cells. The application of this tracer technology can help discover more diverse lactylation-modified proteins in the future and reveal the association of dynamic changes in lactylation modifications with the development of major chronic diseases such as lactate disorders driving inflammation and tumors, suggesting new therapeutic targets. This work was supported by the National Key Research and Development Program of China, the National Natural Science Foundation of China, the XingPharma Scholars Program of China Pharmaceutical University, the Independent Research Project of the State Key Laboratory of Natural Drug Active Component and Drug Efficacy, and the Basic Research Business Fund of Central Universities.
Article Link: https://www.nature.com/articles/s41592-022-01523-1





